Research
Main content
We develop multiscale statistical-dynamical models — from first-principles calculations to coarse-grained kinetic simulations — to understand and predict behaviors in complex chemical and materials systems. Recent work focuses on nano-electrocatalysis, interfacial self-assembly of nanomaterials, and the interplay between nanoscale geometry and reaction kinetics. By bridging the time and length scale gaps between atomic processes and experimental observations, we aim to provide design principles for next-generation functional materials and catalysts.
Representative results are given as follows:
Interfacial-assembly-induced in situ transformation from 1D nanowires to quasi-2D nanofilms
We discovered that well-aligned 1D Te nanowires on a liquid-air interface can spontaneously undergo an oriented attachment process, transforming into quasi-2D nanofilms. Ensemble-averaged energy sampling and Monte Carlo simulations reveal that nanowires maintain their lowest interaction energy at the ordered crystal plane (110) orientation, then rearrange and attach to form the quasi-2D film. This 1D-to-2D transition provides new opportunities for material design at the interface.
 J. Am. Chem. Soc. 146, 19998–20008 (2024).
J. Am. Chem. Soc. 146, 19998–20008 (2024).Plateau–Rayleigh instability in soft-lattice inorganic solids
Plateau–Rayleigh (P-R) instability — the breakup of a fluid cylinder into droplets — has never been observed in conventional inorganic solids due to their rigid lattices. We reported the first solid-state P-R instability in silver-based chalcogenide semiconductors (Ag2E, E = S, Se) featuring soft ionic lattices. The high ionic diffusion coefficients enable conformal core-shell nanowires to self-limitedly morph into periodically coaxial heteronanowires, opening routes toward sophisticated nanostructures at all-inorganic solid-solid interfaces.
 J. Am. Chem. Soc. (2024).
J. Am. Chem. Soc. (2024).Microchemical engineering in a 3D ordered channel enhances electrocatalysis
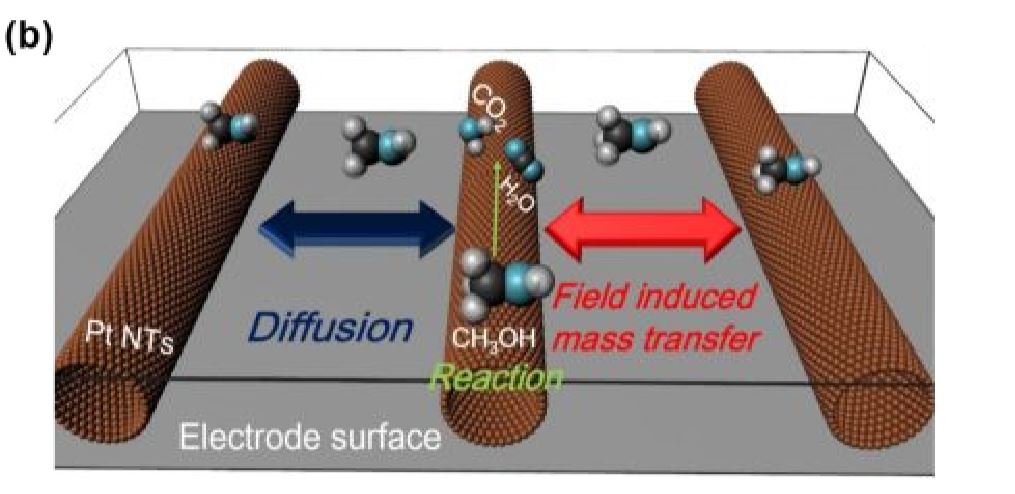
We demonstrated that well-designed nanocatalysts with 3D ordered periodic structures can optimize mass-transport kinetics from bulk electrolyte to the catalyst surface by directional micro-electric fields. The gradient of the local electric field directs reactant flux uniformly to the nearest catalyst site, enabling all reactant molecules to be utilized sufficiently. This concept was confirmed in CO2 reduction, hydrogen evolution, and oxygen reduction across various nanoassemblies.
 J. Am. Chem. Soc. (2021).
J. Am. Chem. Soc. (2021).Proximity-dependent selectivity of CO2 reduction on bifunctional catalysts
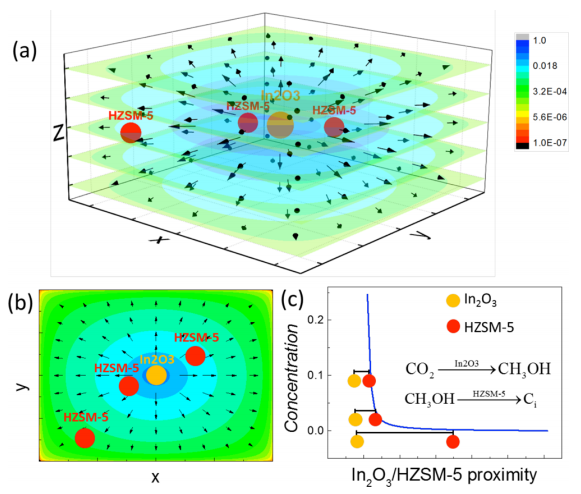
We reveal that the well-known proximity-dependent selectivity of multifunctional catalysts is closely associated with the kinetics involved. Based on reaction-diffusion dynamics together with kinetic Monte Carlo simulation on a coarse-grained model, we found that the diffusion kinetics of the intermediate methanol generated on In2O3 plays a decisive role for the selectivity, resulting in a preferred reaction window to generate favorable liquid fuels with high selectivity.
ACS Catal. 2020, 10, 13518–13523
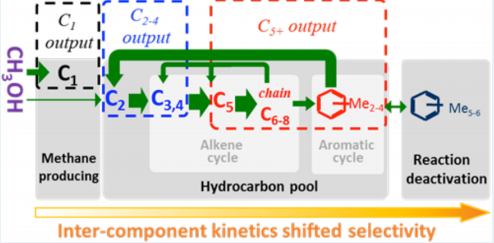
Roughness-enhanced selectivity of carbon monoxide reduction
High roughness has been proved to be an effective design strategy for electrocatalysts. We unraveled the hidden mechanism behind roughness-enhanced selectivity by establishing a comprehensive kinetic model for CORR on catalysts with different roughness factors. We conclude that the roughness-enhanced CORR selectivity is actually kinetically controlled by local electric-field-directed mass transfer of adsorbed species on the electrode surface.
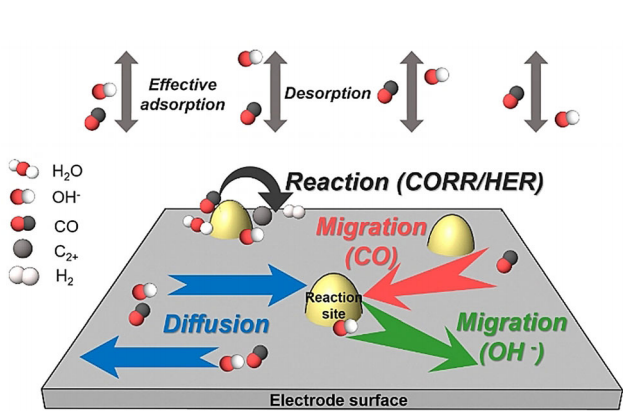
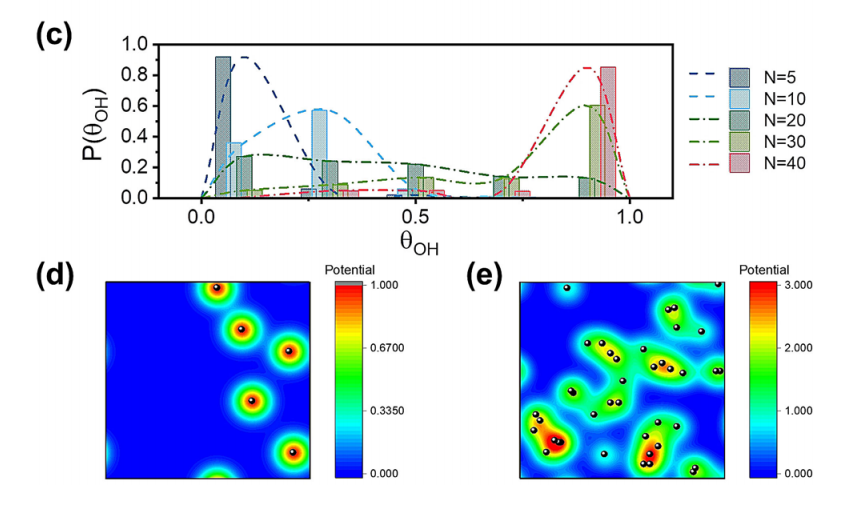
Angew. Chem. Int. Ed. 2020, 60, 1–6
Ordered nanostructure enhances electrocatalytic performance by directional micro-electric field
Well-designed nanocatalysts with periodic structures can optimize kinetics to accelerate mass-transport from bulk electrolyte to the catalyst surface by the gradient of the microelectric field directing uniformly to the nearest catalyst, enabling the boost of electrocatalytic performance. This concept is confirmed in various catalytic systems and nanoassemblies.

Kinetics of tip/roughness-enhanced selectivity of electrocatalytic reduction
A versatile strategy is presented to track the assembly evolution of nanowires in real time. During the interface assembly process, the randomly dispersed NWs gradually aggregate to form small ordered NW blocks and finally are constructed into well-defined NW monolayer driven by the conformation entropy. The assembly mechanism is revealed by thermodynamic analysis and large-scale molecular dynamics simulation.
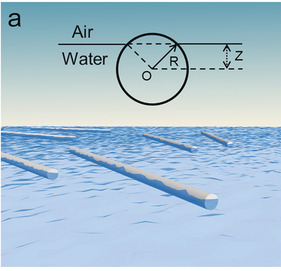
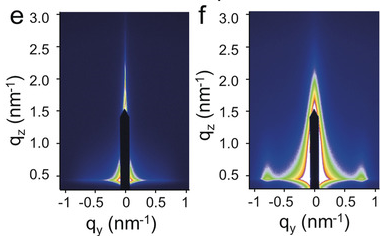
Angew. Chem. Int. Ed. 2018, 57, 8130–8134
Multiscale modeling of nonlinear epitaxial growth of graphene
To bridge the huge time scale separation between atomic processes and experimental kinetics in graphene growth, we developed a multiscale approach combining DFT calculations with kinetic Monte Carlo simulation. Analysis shows that lattice mismatch between graphene and the metal substrate is the origin of the nonlinear growth behavior, and the growth exponent is geometrically determined by the Moiré pattern.