Research
Main content
Biomacromolecules self-organize into membraneless organelles and dynamic condensates through liquid-liquid phase separation (LLPS), a process central to cellular function. We combine coarse-grained molecular dynamics, phase-field modeling, and polymer physics theory to understand how sequence features of intrinsically disordered proteins (IDPs), multivalent interactions, and nonequilibrium driving forces collectively govern LLPS. We also study higher-order chromatin organization and the mechanical properties of protein aggregates linked to neurodegenerative diseases.
Representative results are given as follows:
Sequence-encoded patterning of stickers modulates biomolecular condensate reconfiguration
IDPs drive phase separation via "sticker-and-spacer" interactions. We showed that the mesoscopic patterning of stickers along the IDP sequence — not just their number — critically modulates condensate morphology and reconfiguration. Sparse, homogeneous stickers promote robust aggregation, while proximal sequential organization induces dispersed small clusters. These findings illuminate how sequence-level encoding controls the material properties of biological condensates.
 J. Chem. Phys. 164, 085103 (2026).
J. Chem. Phys. 164, 085103 (2026).Motorized chromosome models of mitotic chromosome folding
During mitosis, chromosomes reconfigure from spherical into rod-like structures. We developed a hybrid motorized chromosome model incorporating two types of "grappling motors" corresponding to condensin I and II. Condensin II facilitates large-scale scaffold formation while condensin I organizes local helical loops. Together they produce the hierarchical helical structure of mitotic chromosomes. The model also explains the formation of structural defects like perversions and entanglements and how topoisomerase action resolves them.
 Nat. Commun. 16, 11085 (2025).
Nat. Commun. 16, 11085 (2025).Molecular mechanism of temperature-dependent phase separation of HSF1
Heat shock factor 1 (HSF1) is the central orchestrator of cell responses to heat shock. We discovered that HSF1 exhibits temperature-dependent LLPS with a lower critical solution temperature behavior, providing a new mechanism for HSF1 activation. Using residue-level coarse-grained simulations combined with NMR experiments, we mapped the molecular interfaces driving phase separation of wild-type HSF1 and its distinct post-translational modification patterns, revealing a species-specific chemical code linking LLPS to physiological temperature control.
 Nat. Chem. Biol. (2025).
Nat. Chem. Biol. (2025).Protein binding domain distribution cooperatively influences protein-RNA LLPS
Using a patchy-particle polymer model, we systematically investigated how the spatial distribution and relative size of protein binding domains (PBDs) influence LLPS in protein-RNA mixtures. We found a nontrivial cooperative dependence: sparsely distributed PBDs favor LLPS for small proteins, while closely packed PBDs facilitate LLPS for larger ones. The former produces multilayer protein clusters around RNA, while the latter favors bilayer dense clusters — providing design rules for engineering biomolecular condensates.
 J. Phys. Chem. B (2025).
J. Phys. Chem. B (2025).Motion transition of active filaments
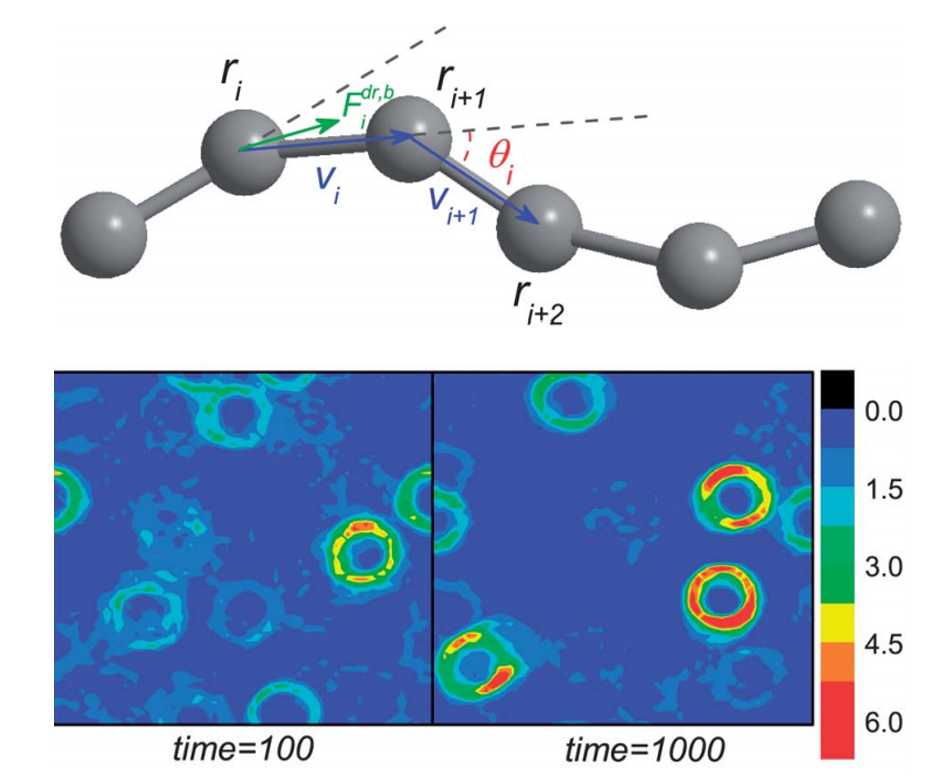
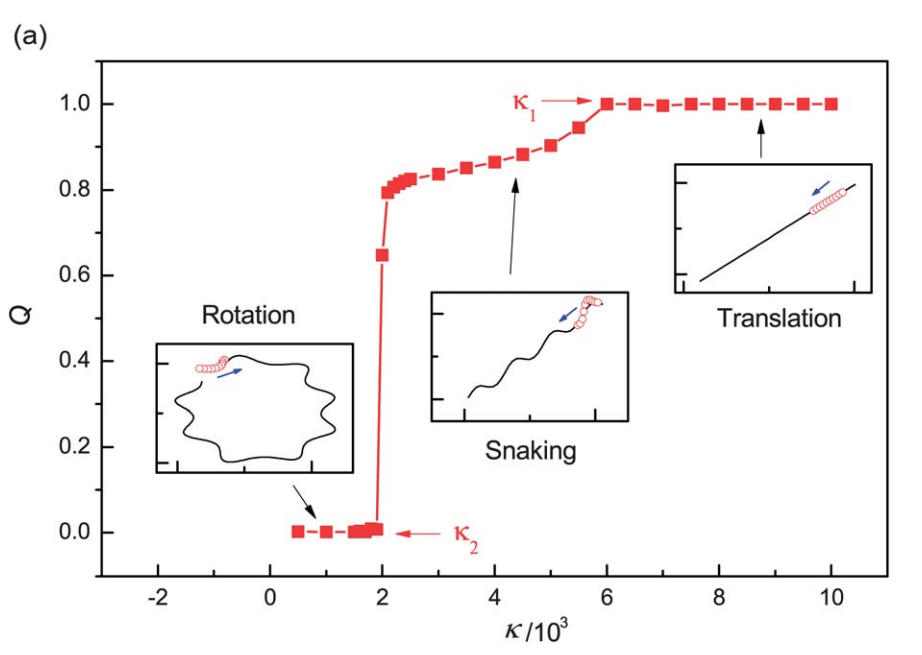
Active filaments can self-organize into fascinating coherently moving structures, such as propagating density waves and vortexes. We find that the filament can show three distinct types of motion — translation, snaking, and rotation — with the variation of the rigidity or active force. Hydrodynamic interaction is not necessary for the rotation or snaking motion, but can enlarge remarkably the parameter regions in which they can occur, providing new insights into the role of HI in collective structures of active systems.
Soft Matter, 2014, 10, 1012–1017
Hydrogen-bond regulation on cytosine dimer assemblies
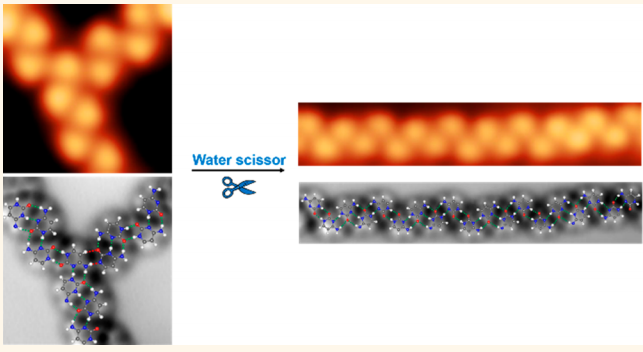
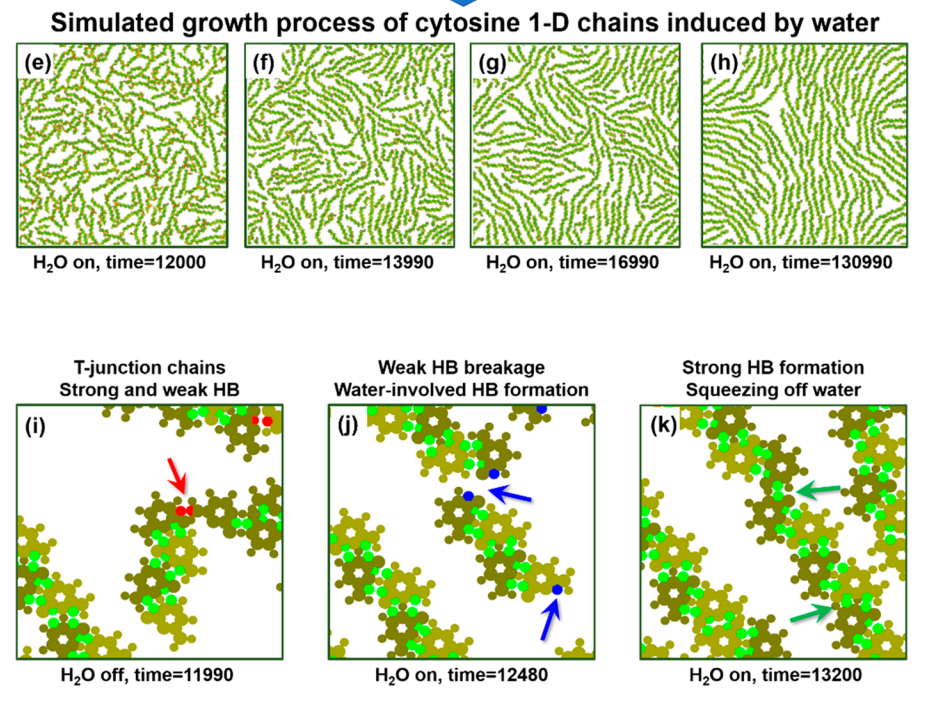
The solvation of one of the DNA bases, cytosine, whose glassy-state network formed on Au(111) contains diverse types of hydrogen-bonded dimer configurations with hierarchical strengths, is investigated. Upon water exposure, a global structural transformation from interwoven chain segments to extended chains occurs. DFT calculation and coarse-grained MD simulation indicate that water molecules selectively break the weak-hydrogen-bonded dimers at T-junctions, while the stable ones within chains remain intact.